OxyCheck is the world’s first in-line measurement system that can check the oxygen content and seal integrity of every single package in a modified atmosphere packaging line.
It was introduced at the International Production & Processing Expo (IPPE) in Atlanta, Georgia, from January 28-30, 2020.
With GEA OxyCheck, every MAP (Modified Atmosphere Package) is non-invasively inspected for oxygen content after sealing on a GEA thermoformer. Up until now, packages had to be damaged by a probe and testing was limited to a small sample coming off the line.
“OxyCheck will be an important add on to our thermoforming equipment. Large customers have already shown interest and are waiting for the new technology. At IPPE we will show our new thermoformer, the GEA PowerPak PLUS together with the GEA OxyCheck. Both have been developed to make the customer’s packaging job easier, which means more efficiency, improved package quality, reduced film consumption, and simple handling, as well as safe process sequences and greater availability,” says Steve McGady, GEA head of equipment sales, slicing and packaging, North America.
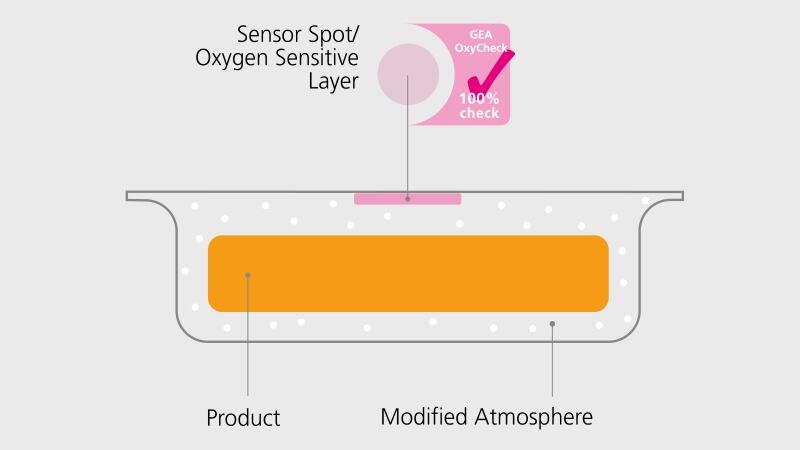
Today, more than half of all retail fresh food – including dairy products – is packaged in a modified atmosphere. During this process, a gas mixture of carbon dioxide and nitrogen is injected into a package prior to sealing. This slows down decay, which extends the shelf life of the packaged product. A small percentage of oxygen remains in the package by virtue of the process, but this may not exceed a threshold. GEA OxyCheck carries out the task of monitoring the residual oxygen content directly during packaging on the GEA PowerPak PLUS thermoformer.
The GEA technology uses a sensor spot made of an FDA approved polystyrene-based fluorescent dye that is printed on the inside of the top film. Similar to an indicator strip, the wavelength of the sensor spot changes depending upon the oxygen content inside the package. Sensor spot readers are installed on the GEA PowerPak PLUS thermoformer after the package has been gas injected and hermetically sealed.
GEA has developed a method for calculating the oxygen concentration based on the light emitted and the temperature. The new measurement system has no effect on either the line speed or the throughput of the GEA PowerPak PLUS.
Up until now, spot checks have been carried out to measure the oxygen content in MAP – which means that less than 0.5% of the packs manufactured are typically tested. Spot checks involve the removal of packs from the line and their inspection through an invasive probe. When a pack does not meet specifications, the line is halted and all packs preceding it up to the last pass are destroyed.
However, even perfectly fine packages are disposed of because they are damaged by the needles during the testing process.
The GEA OxyCheck process is non-invasive and neither the product nor the packaging materials go to waste. And 100% of the manufactured packs are inspected, so only packages that meet all specifications enter the supply chain.
“We also help food manufacturers avoid costly recalls of whole batches, which retailers can demand even if only one pack shows early spoilage,” Volker Sassmannshausen, GEA senior product manager thermoforming, said.
Initially designed for use on the horizontal thermoformers of the PowerPak line, GEA OxyCheck can be installed as an optional feature on the latest generation of PowerPak thermoformer. All existing machines that are equipped for MAP can be outfitted with this monitoring system. The system can then be configured to reflect on-site conditions and customer needs.

