The study analysed changing L. monocytogenes environmental contamination patterns in an Austrian cheese processing facility from 2010 to 2013.
Overall Listeria spp. occurrence rate was 21.9%, dominated by L. monocytogenes (19.5%).
Although susceptible to a range of disinfectants and cleaners, one dominant L. monocytogenes sequence type (ST5) could not be eradicated from drains and floors.
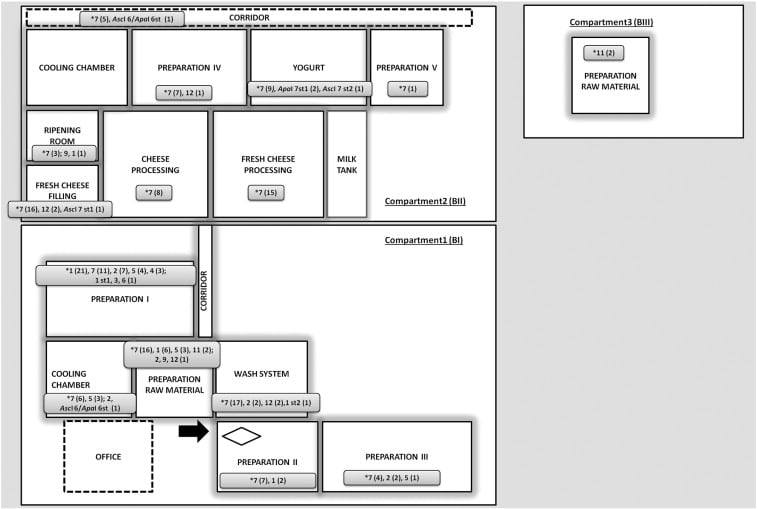
The plant produces fresh cheese produces, ripened brine curd cheese and semi-hard cheeses, all made from pasteurized cow, ewe and goat milk.
It comprised of one compartment for production of fresh cheese products, including rooms for preparation of raw materials and a cooling chamber (BI), an extension building, mainly for dairy processing (yogurt, semi-hard cheese processing, fresh cheese processing) (BII), and one for storage and preparation of raw materials (BIII).
Samples tested
Sampling included non-food contact surfaces (NFCS; drain water, floors, walls, tables, trays, and trolleys), food-contact surfaces (FCS; e.g. fillers, molds, milk vats, slicers; tables, trays, and trolleys), and gloves, work aprons and shoes from dairy staff.
A total of 1,284 environmental samples were tested for the presence or absence of Listeria spp. including L. monocytogenes.
183 drain water and 1101 swab samples were taken from three compartments of the facility.
All samples were transported to the laboratory of the Institute of Milk Hygiene, Milk Technology and Food Science (IMMF) and investigated immediately.
15.3% of the samples were contaminated with L. monocytogenes, 3.4% contained Listeria species mixtures (L. monocytogenes, L. innocua, and L. welshimeri), and 0.9%, mainly floor and drain samples, contained L. monocytogenes mixtures.
The study was complemented by epidemiological tools for strain characterization, recording changes in hygiene management strategies, susceptibility tests of recurrently isolated L. monocytogenes strains to disinfectants and cleaners and survival experiments during storage on the food matrix.
Characterization of L. monocytogenes isolates included genotyping by pulsed-field gel electrophoresis (PFGE) and multi-locus sequence typing (MLST).
Identical L. monocytogenes genotypes were found in the food processing environment (FPE), raw materials and in products.
Sampling phases
FCS swabs from fillers were mainly positive for L. monocytogenes (4.7%) in the first sampling phase.
After improving hygiene management, e.g. strict compartmentalization of the hygiene areas, and staff training, e.g. following to one-way traffic routes and correct usage of hygiene locks, FCS were Listeria spp. negative.
In subsequent monitoring, extensions and reconstructions of the facility took place.
Further testing of NFCS (conveyor belts, transport trolleys, pallets, and tables) resulted in 1.4% of samples positive for L. monocytogenes.
The processing environment (drains, walls, doors and floors) was found to be L. monocytogenes positive during the whole investigation (15.8%).
L. monocytogenes contamination spread from the processing environment in building compartment BI to BII. As floor contamination increased, shoes were frequently found to be positive (48.4%).
The third contamination scenario in the final phase (2012-2013) was reduction to one predominant genotype FCP7 (ST5), with occasional detection of FCP12 (ST204) in the drains and on the floors.
In general, all L. monocytogenes test strains were susceptible to peracetic acid (MIC 250 mg/l), H2O2 (MIC 125 mg/l), Prodesan PE 15 and Citrosan (MIC 500 mg/l), and Rimalkan (MIC 15714 mg/l).
Source: International Journal of Food Microbiology
Online ahead of print, DOI: 10.1016/j.ijfoodmicro.2014.08.001
“L. monocytogenes in a cheese processing facility: Learning from contamination scenarios over three years of sampling”
Authors: I. Rückerl, M. Muhterem-Uyar, S. Muri-Klinger, K.-H. Wagner, M. Wagner, B. Stessl
