The addition of probiotics has grown common in fermented dairy products in the US. With today’s health-conscious and clean label-driven consumer, more brands are prioritizing functional ingredients that support gut health alongside live and active probiotic cultures.
The living microorganisms found in dairy products must be documented and shown to have health benefits in humans when consumed in large amounts, according to the World Health Organization (WHO). But labeling across the probiotics industry is unclear, with room for improvement.
Ingredient weight vs viable count
Gut, immune and digestive system health are major attractions of probiotic dairy products, with dairy yogurts, beverages and cottage cheese snacks leading the category. But most labeling is inconsistent, and the FDA does not require any probiotic quantity details in food products.

Some legacy brands, like Dannon’s Activia, note that each yogurt product contains ‘billions of probiotics’ and also which culture (Bifidobacterium animalis lactis DN-173 010/CNCM I-2494). But others, like Good Culture, only state that its cottage cheese and sour cream products contain ‘live and active cultures’ with no further detail.
Good Culture recently launched a line of wellness probiotic gut shots, and says on its website that they contain “50 billion live and active cultures and 12 live and active strains.” The inconsistencies in probiotic dairy mirror that of probiotic dietary supplements.
Data from the International Probiotics Association (IPA) says that the global probiotic supplements market is now worth $6.07bn, and the global functional/fortified probiotic yogurt category is worth $31.1bn. There are 53 million consumers of probiotics in the US, 58% of them consuming yogurts and 42% consuming supplements.
Current dietary supplement labeling in the US is required to have the milligrams of the product’s active ingredient listed on the nutrition facts panel. But the IPA recently filed a citizen’s petition stating that this doesn’t make sense for probiotics.
The weight of the ingredient is not important, but the count of the viable microorganisms is. The FDA responded with a draft guidance that stated regulations on the topic would not be established or enforced.
The enforcement discretion recommends that probiotics products list the Colony Forming Units (CFU) on the label, and the FDA does not object to it being on the label, but it will not require it.
“We recognize that manufacturers are using a number of different units of measure for probiotics, enzymes, and other dietary ingredients. We need to fully evaluate each unit of measure for dietary ingredients to determine if it is appropriate for use on the Supplement Facts label, and if there are any implications to allowing for the use of such units of measure on the label,” the FDA said.
Genus, species, strain and dose descriptions
Probiotic microbiology consultant and executive science officer for the International Scientific Association for Probiotics and Prebiotics (ISAPP), Mary Ellen Sanders, told DairyReporter that the current casual labeling practices are a concern for the industry.
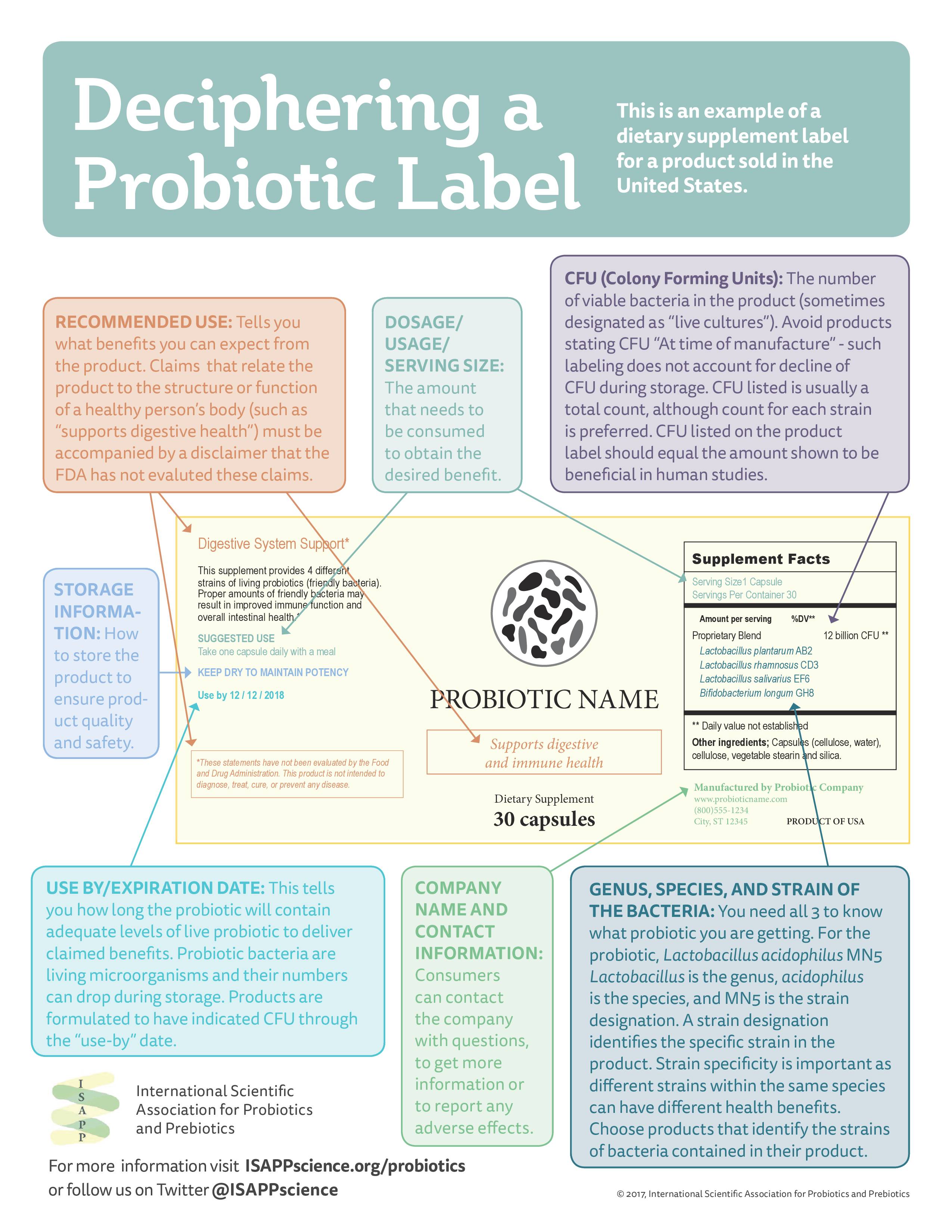
The ISAPP advocates for responsible labeling in probiotics, and says that the CFU count through the end of a product’s shelf life should always be indicated on the package, whether concerning dairy foods or dietary supplements.
“From our point of view, if you’re marketing a dairy product that says ‘contains probiotic’ you should also list the strain of probiotic, or strains that are contained, and the count at the end of shelf life,” Sanders said.
But she believes that the solution rests largely with the industry stepping up and improving labels on their own, rather than struggling with the back-and-forth of getting laws passed and regulations established.
“I don’t feel personally that the FDA has to issue a regulation requiring it, but I do think on a voluntary basis that the industry should do this on their own,” Sanders said.
Beyond the CFU count, ISAPP also thinks that food and supplement packaging should contain a description of the probiotic, including the genus, species, strain and dose of the microorganism.
“More important than supplement vs food is getting an effective probiotic strain at an effective dose,” ISAPP said. “Higher counts are not necessarily better. Levels of live probiotics should be provided through the ‘best by’ date, not time of manufacture.”


